Understanding DMSO and Glucose
in Biologic Preservation
Understanding DMSO and Glucose
in Biologic Preservation
What Is the Goal of Preservation in Biologic Materials?
Preservation methods are used in laboratory and research settings to support the stability of biologic materials during storage and transport.
When biologic materials are stored at ultra-low temperatures, preservation systems are designed to assist with structural integrity during freezing and thawing processes. Different excipients and cryoprotectants may be incorporated depending on the material type and laboratory protocol.
Following thawing, handling procedures and formulation design may influence material characteristics. Preservation strategies are selected based on compatibility with processing methods, storage requirements, and research objectives.
Glucose and other sugars are sometimes included in preservation systems as formulation components. The selection of any preservative system depends on laboratory design considerations and does not imply therapeutic benefit or clinical superiority.
Descriptions provided are educational in nature and do not represent claims of safety, effectiveness, or FDA approval for clinical use.
What Is DMSO?
Dimethyl sulfoxide (DMSO) is an organosulfur compound commonly used as a laboratory solvent and cryoprotectant. It has the ability to dissolve both polar and nonpolar substances and is frequently utilized in research settings for preservation and formulation purposes.
DMSO has also been studied in certain medical contexts and is FDA-approved for limited applications, such as the treatment of interstitial cystitis. Its broader clinical uses remain subject to regulatory evaluation and scientific review.
Safety Considerations in Clinical Contexts
Like many compounds used in laboratory and medical environments, DMSO has been studied for potential side effects depending on concentration, route of administration, and clinical context.
Published literature has reported that exposure to DMSO in medical settings may be associated with a range of reactions, which can vary in severity.
Reported reactions in certain contexts have included:
- Transient odor or taste changes
- Skin irritation or dryness
- Headache or dizziness
- Gastrointestinal discomfort
In some infusion-based settings, additional reactions have been documented in medical literature. The nature and frequency of such reactions depend on multiple clinical variables, including dosing and patient-specific factors.
This information is provided for general educational purposes and does not imply risk associated with any specific product or formulation.
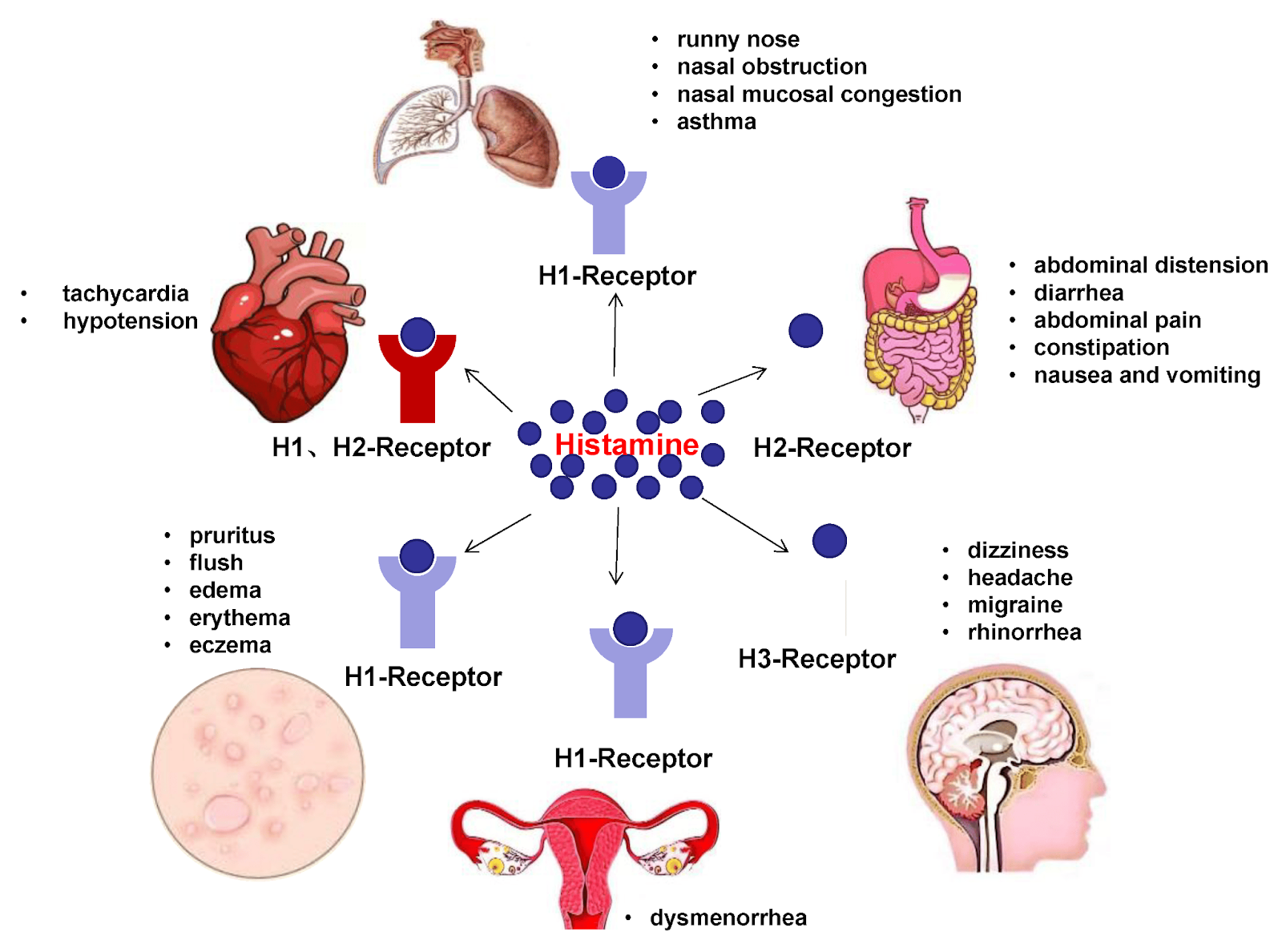
Figure #1: Systemic consequences of mast cell histamine release.
Regulatory Context
DMSO may be used in laboratory preservation systems under defined protocols. The regulatory classification of products containing DMSO depends on intended use, formulation, and jurisdiction.
This page does not compare preservation systems or claim superiority of one approach over another.
Benefits of Glucose-Based Preservation
Educational Overview
GGEVITYY utilizes a glucose-based preservation approach and does not incorporate DMSO in certain formulations.
Preservation methods vary depending on laboratory protocols, material type, and storage objectives. Glucose and other sugars are commonly referenced in scientific literature as excipients that may contribute to stabilization in specific research and cryopreservation contexts.
- 1
Formulation Considerations: Glucose is a naturally occurring carbohydrate frequently used in laboratory and pharmaceutical formulations as a stabilizing excipient. Its inclusion in preservation systems depends on compatibility with specific processing and storage protocols.
- 2
Research Context: Scientific literature has examined various cryoprotectants and excipients, including DMSO and sugar-based alternatives, in laboratory preservation settings. These studies evaluate parameters such as structural integrity and post-processing characteristics under controlled conditions.
- 3
Handling & Storage Objectives: Preservation systems are selected based on laboratory handling requirements, freeze-thaw conditions, and formulation design. The choice of excipient does not imply therapeutic use or clinical superiority.
In conclusion, glucose preservation in natural biologic products offer a safer alternative to DMSO, reducing the risk of adverse reactions while maintaining the viability of the biologic. This is a significant differentiator for GGEVITYY, ensuring that our biologics are of the highest quality, with the top priority being the impact they have on the end user.
Important Notice
Information provided on this page is educational in nature. It is not intended to diagnose, treat, cure, or prevent any disease. Preservation methods described do not imply FDA approval or clinical efficacy. Any clinical decisions must be made by licensed healthcare professionals in accordance with applicable regulations.